|
For i-STAT test cartridge information and intended use, refer to individual product pages or the cartridge information (CTI/IFU) in the i-STAT Support area.Ībbott - A Leader in Rapid Point-of-Care Diagnostics. Check with your local representative for availability in specific markets. Not all products are available in all regions. GDPR Statement | Declaration for California Compliance Law. Any person depicted in such photographs is a model. Photos displayed are for illustrative purposes only. Your use of this website and the information contained herein is subject to our Website Terms and Conditions and Privacy Policy. The products and information contained herewith may not be accessible in all countries, and Abbott takes no responsibility for such information which may not comply with local country legal process, regulation, registration and usage. 
i-STAT CHEM8+ (blue) Cartridge: Now also U.S. 
This website is governed by applicable U.S. i-STAT products alerts and notices for Abbott Point of Care. No use of any Abbott trademark, trade name, or trade dress in this site may be made without the prior written authorization of Abbott, except to identify the product or services of the company. Requests to FDA for agency records should be sent to: Food and Drug Administration Division of Freedom of Information (HFI-35), 5630. Unless otherwise specified, all product and service names appearing in this Internet site are trademarks owned by or licensed to Abbott, its subsidiaries or affiliates. To obtain additional available information, contact FDA. Together, they create the i-STAT System a point-of-care-testing platform that provides healthcare professionals with diagnostic information when and where it’s needed. For i-STAT test cartridge information and intended use, refer to individual product pages or the cartridge information (CTI/IFU) in the i-STAT Support area.Ībbott - A Leader in Rapid Point-of-Care Diagnostics.©2023 Abbott. The i-STAT 1 can connect to multiple data management systems including Abbott Info HQ or AEGISPOC TM - to integrate seamlessly with your Laboratory Information System, Electronic Medical Records or both to help you manage test results, operators and devices. The products and information contained herewith may not be accessible in all countries, and Abbott takes no responsibility for such information which may not comply with local country legal process, regulation, registration and usage. 20, 2021 / PRNewswire / - Abbott (NYSE: ABT) today announced that the U.S. 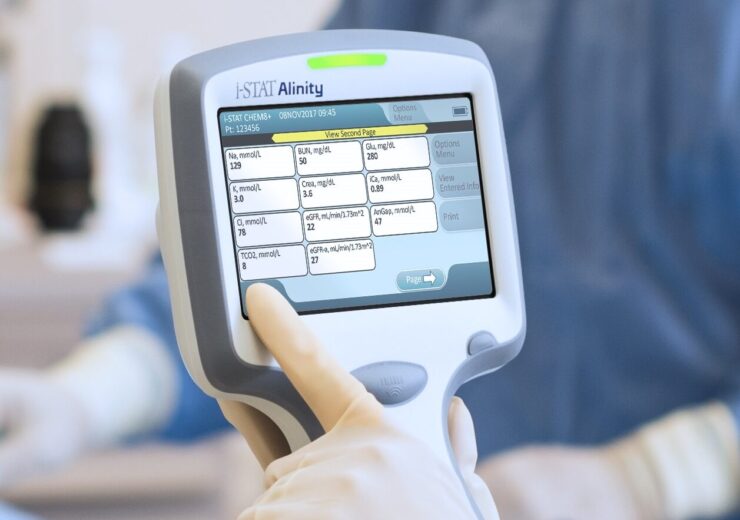
This website is governed by applicable U.S. 
Unless otherwise specified, all product and service names appearing in this Internet site are trademarks owned by or licensed to Abbott, its subsidiaries or affiliates. SKU ABBT-04J60-20 (1 Analyzer) i-STAT Waived Distributor Kit (06F1610, 06F20-03, 06F1101, 06F2391 - downloader should be selected separately if required all items ship in one box) CLIA-waived i-STAT Portable Clinical Analyzer - Blood Gas Test (Abbott Portable Handheld Clinical Analyzers).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
